SAVE ÇÖZÜMLERİ
Katodik
Koruma
Katodik Koruma Sistemleri Tasarımında Uzmanlık
SAVE Mühendislik, teknik hakimiyeti sayesinde verimli projelendirmenin anahtarı olan isabetli analiz süreçleri yürütüyor ve sistem tasarımında tüm elemanların doğru tespiti ile uzun ömürlü çözümler sunuyor.

Kalıcı Koruma
Katodik koruma sistemlerinin analiz ve projelendirme aşamalarında verilen her karar sistemin verimine ve ömrüne etki ediyor. Her detaya gereken özeni gösteren SAVE Mühendislik, sistem tasarımı sürecinde uzun ömürlü, sürdürülebilir bir koruma sağlamayı hedefliyor.

Metal Nasıl Korozyona Uğrar?
Korozyon, metal yüzeyinde oluşan anot ve katot reaksiyonlarının bir arada yürümesi sonucunda meydana gelir.
Bu reaksiyonlar için gereken 3 etken şöyledir:
1. Anot ve katot (İki farklı potansiyelde metal)
2. Elektrolit (tuzlu su çözeltisi)
3. Anot ve katot arasında elektriksel bağlantı
İki farklı potansiyelde metal ifadesi, çelik ve alüminyum gibi tamamen farklı alaşımlar olabileceği gibi genelde mikroskobik ve makroskobik metalürjik farklılıkları olan iki aynı tür metal anlamına gelir.
Örneğin demir yüzeyindeki anot katot reaksiyonları şöyledir:
Anot reaksiyonu: Fe → Fe 2+ + 2 e-
Katot reaksiyonu: ½ O2 + H2O + 2e- → 2OH-
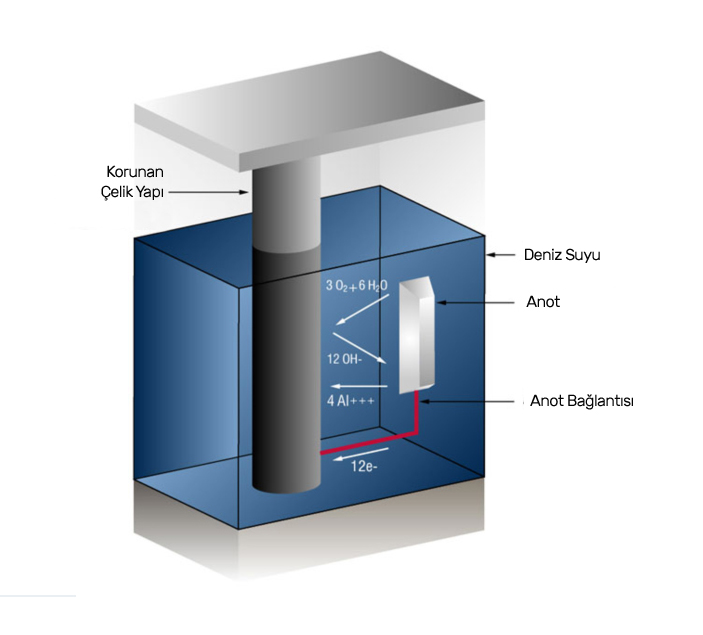
Anot ve katot reaksiyonlarında, elektronlar anottan katoda doğru bağlantı metali üzerinden akar. Katot reaksiyonları gelen bu elektronları kullanarak gerçekleşir. Elektronların katotlarda kullanılmasını engellersek, yani oksijeni buradan uzak tutarsak korozyonu önleriz.
Katodik Koruma, Korozyonu Nasıl Durdurur?
Katodik koruma metallerin korozyonunu kesin olarak önleyen etkili bir koruma yöntemidir. Elektrokimyasal korozyon teorisine dayanan bu yöntemin işleyişi, korunmak istenen metalin elektrokimyasal hücrenin katodu haline getirilip metal yüzeyindeki anot akımlarının giderilmesi şeklinde açıklanabilir. Bunu yerine getirebilecek iki tür koruma sistemi mevcuttur:
1. Galvanik Anotlu Katodik Koruma Sistemleri
2. Dış Akım Kaynaklı Katodik Koruma Sistemleri
1. Galvanik Anotlu Katodik Koruma Sistemleri:
Kurban Anot Yöntemi olarak da adlandırılan Galvanik Anot Yöntemi, korumak istediğimiz metalin korozyona uğraması yerine başka bir metalin feda edilmesi prensibiyle çalışan bir sistemdir.
Bu sistemde, korozyondan korumak istenen metale kendinden daha aktif yani daha negatif bir potansiyeli olan başka bir metal bağlanır ve bu sayede katot reaksiyonları için gerekli olan elektronlar galvanik anottan sağlanır. Bu anotlar çinko, alüminyum ve magnezyum gibi alaşımlardan tercih edilir.
Katodik koruma sistemi tasarlanırken anotların, yapıyı korumak için yeterli akımı boşaltmak için doğru şekle ve yüzey alanına sahip olacak şekilde boyutlandırılmalıdır ve bu akımı boşaltırken istenen ömrü sürdürecek yeterli ağırlığa sahip galvanik anotlar kullanılmalıdır. Koruma sisteminin, akım ihtiyacını azaltmak için korunmak istenen metal yüzeyinin uygun bir malzeme ile kaplanarak etkinliği artırılabilir.

2. Dış Akım Kaynaklı Katodik Koruma Sistemleri:
Dış akım kaynaklı katodik koruma metale dıştan bir doğru akım uygulanarak yapılır. Şebekeden alınan alternatif akım bir transformatör redresör sisteminden elde edilen doğru akıma dönüştürülür ve eksi ucu korunacak olan metale, artı ucu da bir yardımcı anota bağlanır. Yardımcı anot olarak, yüksek potansiyele gerek duymadan istenen akımı verebilen ve uzun süre parçalanmadan dayanabilen çeşitli metaller kullanılabilir.
İlk yatırım masrafları galvanik anotlu sisteme göre daha düşük olsa da işletme maliyetleri yüksektir. Elektrolit direncinin yüksek olması halinde doğru akım potansiyeli trafo ünitesinde ayarlanabilir. Bu işlem manuel yapılabildiği gibi katodik koruma devresine sabit bir referans elektrot konularak otomatik olarak da yapılabilir.
Save Çözümleri
Katodik Korumada
Neden SAVE Mühendislik?
Deniz Yapılarında Katodik Koruma
Deniz yapılarında katodik koruma sistemlerinin tasarlanması ve uygulanması için SAVE Mühendislik, Norse ve Yeke firmaları ile oluşturduğu bir konsorsiyum olan YNS Corrosion markası ile hizmet vermektedir.

Haber bültenimize kaydolun
SAVE Mühendislik'in haber bültenlerini almak, yeni ürün ve projelerinden haberdar olmak için abone olun.
